Contact us
Call us at
Available 6:00 AM – 5:00 PM (PST) Business Days
Download
Download Manuals, Datasheets, Software and more:
Feedback
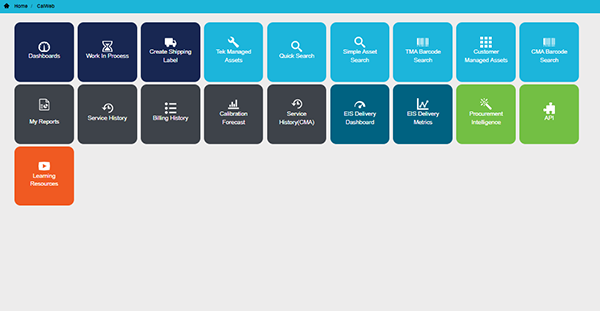
Manage It All From Here
The CalWeb online portal frees you from the manual management of your entire calibration program. Simplify workflows, eliminate overdue calibrations, and streamline audit compliance with CalWeb. With access to CalWeb data and tools, you’ll save time and reduce calibration program complexity. Every day, thousands of Tektronix calibration service customers in mission-critical industries rely on CalWeb for audit compliance and increasing engineering uptime.
With CalWeb, you can:
- Efficiently prepare for service
- Schedule service and monitor it with ease
- Analyze and optimize your calibration program
- Be prepared for audits without the stress
CalWeb Options
CalWeb Essential
- Provides all the essential tools to make global service management easy, efficient, and stress-free for your team
- Included as part of Tektronix service for large equipment fleet customers
- To learn if your company's equipment fleet qualifies for CalWeb Essential to be included with your standard calibration service contact sales
CalWeb Ultra
- All the goodness of CalWeb Essential plus valuable features such as:
- Asset Management: Conveniently manage all your assets that are not service by Tektronix
- Out of Tolerance Case Management: Case Management: track OOT events as you investigate them to resolution
- API: Programmatically integrate your IT application with CalWeb
- Bonded Assets: Easily track preventative maintenance on your assets serviced by Tektronix
- Easily upgrade from within CalWeb Essential

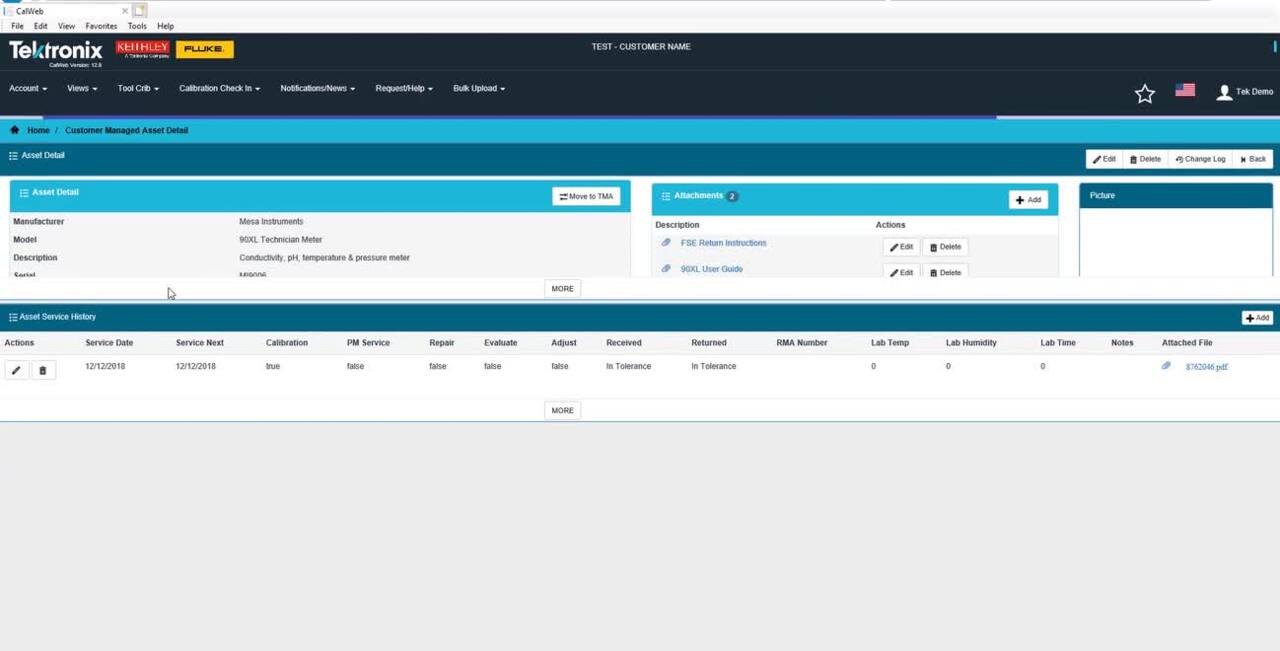
Efficiently Prepare for Service
Store all your asset information in this simple, configurable tool to make it easy to manage your program, from anywhere.
- Know what’s due for calibration, and when, with auto notifications and reporting
- Generate a quote request and view received quotes
- Understand your current and forecasted costs
- Create barcode stickers for all your equipment, setting yourself up for easy information access
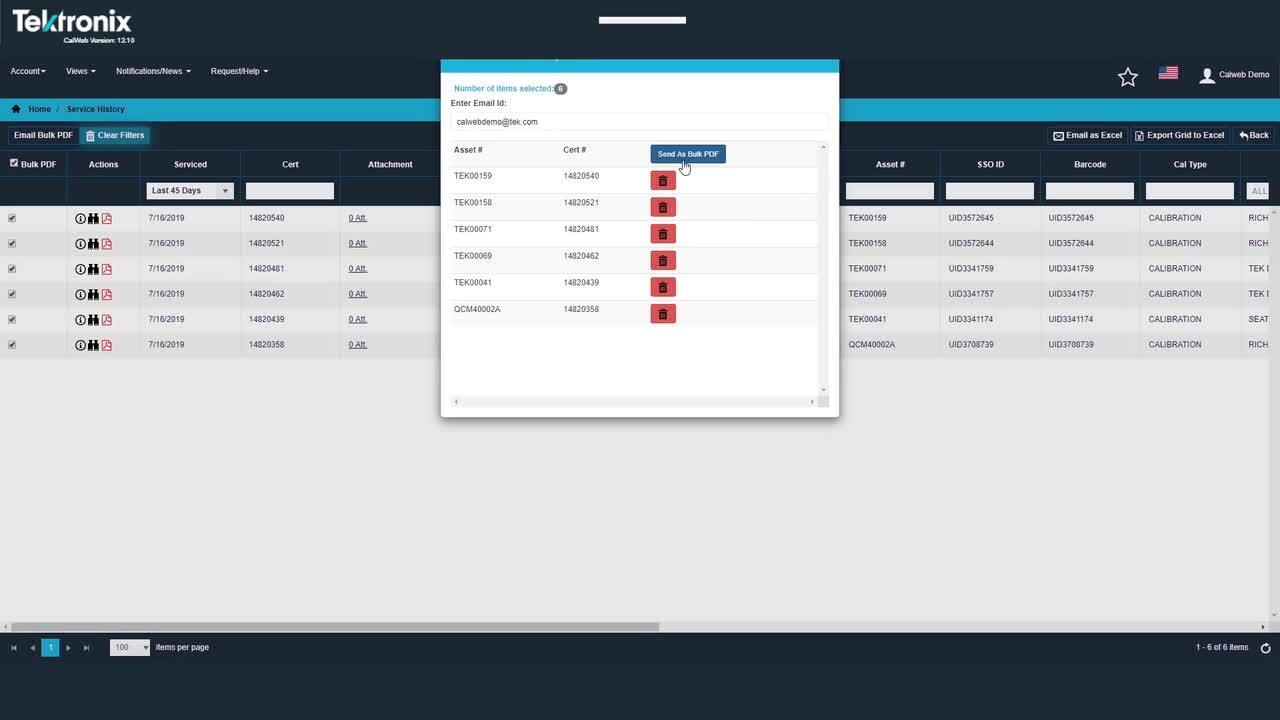
Smooth Service Ordering and Tracking
Use the portal to schedule service and track your units that are in for calibration service, whether your equipment is serviced onsite, at a local lab, or at the Tektronix factory. The dashboard provides instant visibility into your program.
- Order and schedule calibration service online
- Check assets in and out with ease, using barcode scanning
- Generate any necessary documents – shipping labels, packing list, etc.
- Receive updates on calibrations in process
- Communicate with technicians about your assets
- Receive Out of Tolerance notifications and other calibration service results
- Case management for Out of Tolerance events
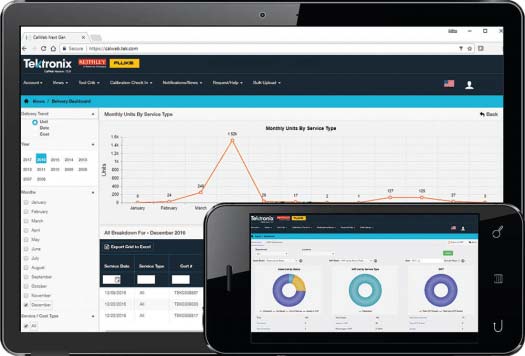
Analyze and Optimize Your Program for Efficiency
CalWeb’s reporting tool makes it easy for you to understand the important trends of your calibration service program.
- Utilize the included standard reports, including calibrations due, work in process, and delivery metrics
- Satisfy your company’s internal metrics by creating custom reports
- Analyze your service history – which units require calibration more often, which units are aging out?
- Analyze your billing history – which units are costing the most, or might need replacement? What is the billing pattern from your calibration service provider?
Pass Audits with Ease
When you manage your program with CalWeb, everything you need for audit compliance is stored at your fingertips for instant access.
- Scan the equipment barcode or use the advanced search function to easily find your equipment details, and access equipment certificates and datasheets
- Instantly produce audit logs, asset history, service history, and payment history on demand

Get to know CalWeb
Get to know CalWeb
- CalWeb Overview
- CalWeb Quick Start
- Manage and View Your Assets
- View Asset Service History
- View Work in Process
- Maintain Audit Compliance
- Instant Visiblity
- CalWeb My Reports
- Customer Managed Assets