Contact us
Call us at
Available 6:00 AM – 5:00 PM (PST) Business Days
Download
Download Manuals, Datasheets, Software and more:
Feedback
A top priority for medical device manufacturers is the health and safety of patients, and the accuracy and precision of the instruments used in production is of the utmost importance. We have decades of experience managing calibration programs in highly regulated industries and provide innovative solutions to optimize even the most complex asset pools. Our skilled technicians can help you stay in compliance with ISO 13485 and 21 CFR parts 11 and 820.
Quality Calibration Program Management
Tektronix has experience managing calibration programs for some of the largest medical device manufacturers in the United States. We offer both traceable and ISO/IEC 17025 accredited calibrations and can help set up calibration programs or provide ongoing business reviews to ensure your company's success. Requirements for staying compliant with ISO 13485 and 21 CFR part 820 are written into the Tektronix quality manual, giving you peace of mind that your company's equipment is being calibrated to the necessary standard. Equipment can be calibrated onsite or in one of our service centers nationwide.

Asset Management
Whether you have multiple brick-and-mortar locations or an army of field service technicians, we can help you manage your equipment at each phase of its lifecycle.
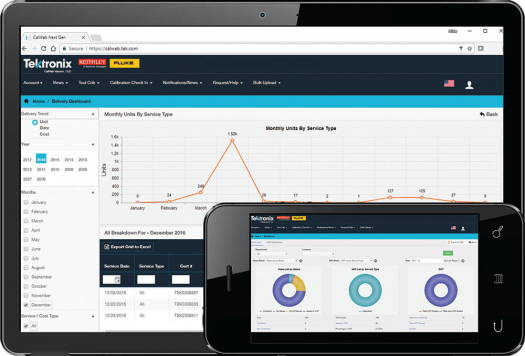
CalWeb
Managing your calibration program with CalWeb simplifies audits and inspections with quick and easy access to your instruments' service history. CalWeb is validated regularly to meet the requirements of 21 CFR Parts 11 and 820.
- Get an organization-wide view of your asset pool
- Store calibration certificates and service records
- Support out-of-tolerance investigations
- Ensure compliance using flexible, customizable reporting
Field Services Management
Tektronix supports the top global medical device companies with one of the country's largest network of service centers. We are well-suited to provide logistical support to field service organizations and companies with multiple locations.
- Lessen your administrative burden by consolidating the number of vendors required to manage assets
- Ensure your technician's equipment is never past-due for calibration
- Eliminate downtime with advanced replacement programs
- Utilize flexible solutions from Tektronix to handle the procurement of consumables and new test equipment
- Track all your assets in CalWeb allowing for ongoing optimization of your asset management processes

Accredited Calibration Services
Our medical test equipment calibration services meet demanding FDA, cGMP, ASTM and CLIA guidelines with flexible delivery options. These include onsite calibration at your facility or in our service centers. Medical equipment calibration services include all the standard metrology capabilities, plus:
- Electrical Safety Analyzer Calibration
- Patient Simulators
- Gas Flow Analyzers
- SPO2 & NIBP Simulators
- EKG Electrode Impedance
- Centrifuges
- Ventilator Tester
- Temperature Simulators, Fluid Warming Systems
- Autoclaves
- Infusion Pumps
- RF / High Frequency Calibration
- Pipette Calibration
- Torque Calibration
- Pressure Calibration
- Temperature / Humidity Calibration
- Electronic Calibration
- And much more!
Tips for Staying in Compliance with 21 CFR 820 and ISO 13485
Learn about the key differences between the two standards and a get a quick guide to choosing a calibration provider that can help you build an ironclad calibration program.
Download Now





